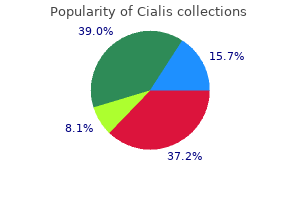
"Buy discount cialis 10mg online, erectile dysfunction in diabetes mellitus ppt".
By: J. Shawn, M.S., Ph.D.
Co-Director, Florida Atlantic University Charles E. Schmidt College of Medicine
The cause is unknown; many cases appear to erectile dysfunction pump how do they work safe cialis 10mg follow benign viral infections or Campylobacter jejuni infection erectile dysfunction doctors in el paso tx discount 5mg cialis amex. Most patients recover spontaneously erectile dysfunction prevalence cheap cialis 10 mg on-line, but as many as one in six may become unable to walk or may develop respiratory failure requiring ventilatory support. The response to therapy is inferior in pa- tients who remain untreated for several weeks. Myeloma light chains may be toxic to renal tubular epithelium and cause renal failure in up to 10% of cases. Homozygous hypercholesterolemia, a rare disorder of the receptor for low-density lipoproteins, results in severe premature atherosclerosis and early death from coronary artery disease. Some patients with heterozygous hypercholesterolemia also develop high levels of cholesterol and are at increased risk for developing premature atherosclerotic heart disease. Treatment is usually performed every 1 to 2 weeks, and cholesterollowering drugs are generally employed simultaneously. Introduction to the third special issue: Clinical applications of therapeutic apheresis. Three different schedules of low-density lipoprotein apheresis compared with plasmapheresis in patients with homozygous familial hypercholesterolemia. Guidance for industry: Recommendations for collecting red blood cells by automated apheresis methods. Photopheresis Photopheresis is a technique that involves the treatment of patients with psoralens, the separation of lymphocytes by apheresis, and treatment of the cells with ultraviolet radiation. Increased granulocyte yields obtained with an oral three-dose prednisone premedication schedule (abstract). Adverse reactions associated with mobile therapeutic apheresis; analysis of 17,940 procedures. Prevention of citrate reactions during therapeutic plasma exchange by constant infusion of calcium gluconate with the return fluid. Transfusion-related acute lung injury during plasma exchange: Suspecting the unsuspected. Vascular erosion caused by a double-lumen central venous catheter during therapeutic plasma exchange. Allergic reactions in healthy plateletpheresis donors caused by sensitization to ethylene oxide gas. Cellular source of serum lactate dehydrogenase elevation in patients with thrombotic thrombocytopenic purpura. Antibodies to von Willebrand factor-cleaving protease in acute thrombotic thrombocytopenic purpura. Cryosupernatant as replacement fluid for plasma exchange in thrombotic thrombocytopenic purpura. Erythrocytapheresis can reduce iron overload and prevent the need for chelation therapy in chronically transfused pediatric patients. Randomized trial of plasma exchange, intravenous immunoglobulin, and combined treatments in 55. Intravenous immunoglobulin treatment in patients with chronic inflammatory demyelinating polyneuropathy. Plasma exchange in polyneuropathy associated with monoclonal gammopathy of undetermined significance. Plasma exchange treatment in rapidly progressive glomerulonephritis associated with anti-neutrophil cytoplasmic autoantibodies. Treatment of polyarteritis nodosa related to hepatitis B virus with short-term steroid therapy associated with antiviral agents and plasma exchanges: A prospective trial in 33 patients J Rheumatol 1993;20:289-98. The Prosorba column for treatment of refractory rheumatoid arthritis: A randomized, double-blind, sham-controlled trial. Familial hypercholesterolemia regression study: A Randomized trial of low-density-lipoprotein apheresis. Protein A columns for the treatment of patients with idiopathic thrombocytopenic purpura and other indications. Treatment of refractory thrombotic thrombocytopenic purpura with protein A immunoadsorption. Although the scope and characteristics of donor tests changed with the release of new tests and the advent of new regulatory requirements, the intent of donor testing remains constant: to enhance the safety of the blood supply. This chapter presents the general principles that apply to testing and labeling donor blood, and it provides a description of the specific tests that are required or done voluntarily at most blood banks on each donation.
Syndromes
- Shampoo less frequently -- perhaps only once or twice a week
- If necessary, take an antihistamine, or apply creams that reduce itching.
- Brain CT scan
- Injection drug users who share needles
- Dry, cool skin
- Headache
- Pain that gets worse over time
- Change in development
Interferon gamma decreases hepatic stellate cell activation and extracellular matrix deposition in rat liver fibrosis impotence from prostate surgery buy generic cialis 2.5mg line. However erectile dysfunction treatment raleigh nc order cialis with a visa, immunotherapy is not successful in all cancer patients due to erectile dysfunction drug warnings generic cialis 20 mg with mastercard both intrinsic non-responsiveness and adaptive resistance. Developing predictive biomarkers and understanding mechanisms of resistance are major goals of the immuno-oncology community. Identification of a novel human memory T cell population with the characteristics of stem-like chemo-resistance. In this work, we carefully examined the specificity and robustness of our customized technology for immunopeptidomic workflow. These results directly imply the opportunity of exploiting microfluidic-based strategy in immunopeptidomic platform and in last analysis in personalized immunopeptidome analysis from individual tumor biopsy to design personalized cancer therapeutic vaccines. Approaches to treat immune hot, altered and cold tumours with combination immunotherapies. Recent efforts to design personalized cancer immunotherapies use predicted neoantigens. The resulting somatic variants of interest are then annotated to predict protein sequence changes and to infer possible neoantigenic peptides. Individual neoantigenic peptides are selected by sliding an amino acid window (usually 811-mers) across the variant position to consider each possible register. Because the accuracy of neoantigen identification has important implications for many clinical trials and studies of basic cancer immunology, there is a need for patient-specific inclusion of proximal variants to address this previously oversimplified assumption in the identification of neoantigens. We evaluated somatic variants from 430 tumors to understand how proximal somatic and germline alterations change the neoantigenic peptide sequence and also affect neoantigen binding predictions. Of these somatic variants, 5% had one or more in-phase missense proximal variants. Our methodology drastically reduces the handling and the timing required for the peptide isolation compared to the reported standard Methods in the field. This in silico sequence analysis method aids in each component essential to the vaccine design process. Keywords: immunogenomics, neoantigens, personalized cancer vaccines, epitope prediction. A neoantigen fitness model predicts tumour response to checkpoint blockade immunotherapy. The relationship between class I binding affinity and immunogenicity of potential cytotoxic T cell epitopes. Insertion-and-deletion-derived tumour-specific neoantigens and the immunogenic phenotype: a pan-cancer analysis. A dendritic cell vaccine increases the breadth and diversity of melanoma neoantigen-specific T cells. These modifications in the levels of antigen expression were transient and did not last more than two weeks after the removal of the cytokines. Healthy individuals have T-cell and antibody responses to the tumor antigen cyclin B1 that when elicited in mice protect from cancer. B164 / Identification of breast cancer neoantigens exposed by radiation therapy Claire Lhuillier (Weill Cornell Medicine), Nils Rudqvist (Weill Cornell Medicine), Takahiro Yamazaki (Weill Cornell Medicine), Lorenzo Galluzzi (Weill Cornell Medicine), Sandra Demaria (Weill Cornell Medicine). Recent studies have highlighted the key role of neoantigens generated by somatic non-synonymous mutations in tumor response to immunotherapy [1]. Epidemiological studies have found that acute febrile infections are associated with a greatly reduced life-time risk of many types of cancer (1, 2). Glioblastoma is the most common and most malignant primary brain tumor with a poor prognosis despite surgery and chemo-radiotherapy. A major challenge is the fact that tumor cells infiltrate the healthy brain tissue, precluding complete surgical resection. Among these, immunotherapy receives increasing attention, also because glioblastoma is paradigmatic for cancer-associated immunosuppression. Interestingly, 2, 064 ligands were shared with cell lines and 3, 754 were presented on at least 2 glioblastoma samples. On the source protein level 239 glioblastoma exclusive proteins were identified; among them 54 were also present in cell lines and 178 were presented on at least 2 glioblastoma samples. Interestingly, 3, 420 glioblastoma-exclusive peptides were presented on at least 2 glioblastoma samples.
Each committee has a written charter outlining its duties and responsibilities erectile dysfunction doctor in delhi order 2.5 mg cialis with visa, and is led by a Board-elected Chairman erectile dysfunction at the age of 20 order 20 mg cialis with amex. Any Board member may request a Board or committee meeting erectile dysfunction education 5 mg cialis for sale, and the inclusion of an agenda item. Before meetings, Board members receive materials to help them prepare the discussions and decision-making. Vice Chairman Enrico Vanni has been the independent, non-executive Vice Chairman since February 22, 2013. Vanni: · Leads the Board in case and as long as the Chairman is incapacitated · Chairs the sessions of independent Board members, and leads independent Board members if and as long as the Chairman is not independent · Leads the yearly session of the Board members evaluating the performance of the Chairman, during which the Chairman is not present 98 Novartis Annual Report 2017 Board meetings the Board has meetings with Executive Committee members, as well as private meetings without them. Because all Board members are independent, no separate meetings of the independent Board members were held in 2017. Subject to additional special meetings, the Board and Board committee meetings take place in January, April, June, August, October and December. Typically these meetings last two days, with the first day allocated to Board committee meetings, and the second day allocated to the meeting of the full Board. Occasionally, other members of management and/ or external advisors are invited to attend and/or present a specific topic at a Board meeting. The independent advisor of the Compensation Committee is regularly invited to attend portions of the meetings of the Compensation Committee. For more information, see Information and Control Systems of the Board vis-а-vis Management, beginning on page 100, and our Compensation Report, beginning on page 118. Daniel Vasella have been appointed Honorary Chairmen in recognition of their significant achievements on behalf of Novartis. An independent Board member is one who is independent of management and has no business or relationship that could materially interfere with the exercise of objective, unfettered and independent judgment. Only with a majority of Board members being independent can the Board fulfill its obligation to represent the interests of shareholders, being accountable to them and creating sustainable value through the effective governance of Novartis. Accordingly, Novartis established independence criteria based on international best practice standards as outlined on the Novartis website. For members of the Audit and Compliance Committee and the Compensation Committee, even stricter rules apply. For this assessment, the committee considers all relevant facts and circumstances of which it is aware not only the explicit formal independence criteria. This includes an assessment of whether a Board member is truly independent, in character and judgment, from any member of senior management and from any of his/her current or former colleagues. In its meeting on December 14, 2017, the Board determined that all of its members are independent. Mandates outside the Novartis Group According to article 34 of the Articles of Incorporation ( Chairmanships of the boards of directors of other listed companies count as two mandates. Mandates in different legal entities that are under joint control are deemed one mandate. As part of this process, each Board member completes a questionnaire on the performance and effectiveness of the Board and the Chairman, and on his/her committees, which lays the groundwork for a qualitative review led by the Chairman. The Chairman has discussions with each Board member and then with the entire Board. In 2017, an independent performance and effectiveness evaluation of the Board and its committees, including an assessment of individual Board members, was conducted by the independent expert company Egon Zehnder. Information and control systems of the Board vis-а-vis management the Board ensures that it receives sufficient information from the Executive Committee to perform its supervisory duty and to make decisions that are reserved for it. Information on management Board members complete questionnaire Board committees Results are collected and evaluated by Board office Chairman has discussion with each Board member Full Board discusses results and agrees on actions Content and results the performance review examines the performance and effectiveness, strengths and weaknesses of individual Board members and of the full Board and each Board committee. This review covers topics including Board composition; purpose, scope and responsibilities; processes and governance of the Board and its committees; meetings and pre-reading material; team effectiveness; and leadership and culture. Board committees regularly meet with management and, at times, external consultants to review the business, better understand applicable laws and policies affecting the Group, and support the Board and management in meeting the requirements and expectations of stakeholders and shareholders. Additionally, the heads of Internal Audit, Financial Reporting & Accounting, Integrity and Compliance, and Quality, as well as the Head of the Global Business Practices Office, report on a regular basis to the Audit and Compliance Committee. For each quarterly and annual release of financial information, the Disclosure Review Committee is responsible for ensuring the accuracy and completeness of disclosures. The Audit and Compliance Committee reviews decisions made by the Disclosure Review Committee before the quarterly and annual releases are published.
Supplier Qualification Critical supplies and services must be qualified on the basis of defined require- ments erectile dysfunction ginseng purchase cialis cheap. Similarly erectile dysfunction treatment boots purchase cialis 20 mg with amex, the supplier should be qualified to erectile dysfunction doctors in maine order cialis 5mg on line ensure a reliable source of materials. The facility should clearly define requirements or expectations for the suppliers and share this information with staff and the supplier. The ability of suppliers to consistently meet specifications for a supply or service should be evaluated along with performance relative to availability, delivery, and support. Examples of factors that could be considered to qualify suppliers are: Licensure, certification, or accredita tion. A list of approved suppliers should be maintained, including both primary suppliers and suitable alternatives for contingency planning. Critical supplies and services should be purchased only from those suppliers who have been qualified. Documented failures of supplies or suppliers to meet defined requirements should result in immediate action by the facility. These actions include notifying the supplier, quality oversight personnel, and management with contracting authority, if applicable. Agreements Contracts and agreements define expectations and reflect concurrence of the parties involved. Blood banks and transfusion services should maintain written contracts or agreements with outside suppliers of critical materials and services such as blood components, irradiation, compatibility testing, or infectious disease marker testing. The outside supplier may be another department within the same facility that is managed independently, or it may be another facility (eg, contract manufacturer). The contracting facility assumes responsibility for the manufacture of the product; ensuring the safety, purity, and potency of the product; and ensuring that the contract manufacturer complies with all applicable product standards and regulations. Both the contracting facility and the contractor are legally responsible for the work performed by the contractor. It is important for the blood bank or transfusion service to participate in the evaluation and selection of suppliers. They should review contracts and agreements to ensure that all aspects of critical materials and services are addressed. Examples of issues that could be addressed in an agreement or a contract include: responsibility for a product or blood sample during shipment; the responsibility of the supplier to promptly notify the facility when changes that could affect the safety of blood, components, or patients have been made to the materials or services; and the responsibility of the supplier to notify the facility when Receipt, Inspection, and Testing of Incoming Supplies Before acceptance and use, critical materials, such as reagents and blood components, must be inspected and tested (if necessary) to ensure that they meet specifications for their intended use. Corrective action may include returning the material to the vendor or destroying it. Receipt and inspection records provide the facility with a means to trace materials that have been used in a particular process and also provide information for ongoing supplier qualification. Equipment Management Equipment that must operate within defined specifications to ensure the quality of blood, components, tissues, and services is referred to as "critical" equipment 1(p4) Critical equipin the quality system. Activities designed to ensure that equipment performs as intended include qualification, calibration, maintenance, and monitoring. When selecting new equipment, it is important to consider not only the performance of equipment as it will be used in the facility, but also any supplier issues regarding ongoing service and support. There should be a written plan for installation, operational, and performance qualification. After installation, there must be documentation of any problems and the follow-up actions taken. Recalibration and requalification may be necessary if repairs are made that affect the critical operating functions of the equipment. Recalibration and requalification should also be considered when existing equipment is relocated. The facility must develop a mechanism to uniquely identify and track all critical equipment, including equipment software versions, if applicable. Maintaining a list of all critical equipment helps in the control function of scheduling and performing functional and safety checks, calibrations, preventive maintenance, and repair. The equipment listing can be used to ensure that all appropriate actions have been performed and recorded.
Cheap 10mg cialis. My Story TURP after a proceedure.

