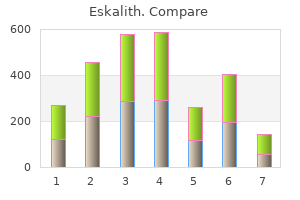
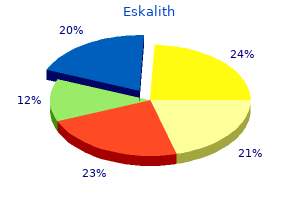
"Purchase eskalith, anxiety ebola".
By: P. Sigmor, M.A., Ph.D.
Deputy Director, Texas Tech University Health Sciences Center Paul L. Foster School of Medicine
Their physical association links the reactions in proper sequence without the release of intermediates depression symptoms recovery buy discount eskalith 300mg. Wernicke-Korsakoff anxiety or adhd purchase 300mg eskalith otc, an encephalopathy-psychosis syndrome due to depression symptoms test online buy eskalith online from canada thiamine deficiency, may be seen with alcohol abuse. Regulation of the pyruvate dehydrogenase complex: Covalent modifications by the two regulatory enzymes that are part of the complex alternately activate and inactivate E1. This enzyme deficiency results in an inability to convert pyruvate to acetyl CoA, causing pyruvate to be shunted to lactate via lactate dehydrogenase (see p. Symptoms are variable and include neurodegeneration; muscle spasticity; and, in the neonatal onset form, early death. The gene for the subunit is X linked, and, because both males and females may be affected, the deficiency is classified as X-linked dominant. This aldol condensation has an equilibrium far in the direction of citrate synthesis. Isomerization of citrate Citrate is isomerized to isocitrate by aconitase (aconitate hydratase), an Fe-S protein (see Figure 9. Oxidative decarboxylation of -ketoglutarate the conversion of -ketoglutarate to succinyl CoA is catalyzed by the -ketoglutarate dehydrogenase complex, a protein aggregate of multiple copies of three enzymes (Figure 9. The equilibrium of the reaction is far in the direction of succinyl CoA, a highenergy thioester similar to acetyl CoA. Cleavage of succinyl coenzyme A Succinate thiokinase (also called succinyl CoA synthetase, named for the reverse reaction) cleaves the high-energy thioester bond of succinyl CoA (see Figure 9. Hydration of fumarate Fumarate is hydrated to malate in a freely reversible reaction catalyzed by fumarase (fumarate hydratase; see Figure 9. Oxidation of malate Malate is oxidized to oxaloacetate by malate dehydrogenase (Figure 9. The most important of these regulated enzymes are those that catalyze reactions with highly negative G0: citrate synthase, isocitrate dehydrogenase, and -ketoglutarate dehydrogenase complex. The central nervous system is particularly affected in this X-linked dominant disorder. Lipoic acid is an intermediate acceptor of the acetyl group formed in the reaction. Pyruvate dehydrogenase complex catalyzes an irreversible reaction that is inhibited when the decarboxylase component is phosphorylated. Administration of thiamine is expected to reduce his serum lactate level and improve his clinical symptoms. Decreased activity of the complex decreases production of acetyl coenzyme A, a substrate for citrate synthase. During a prolonged fast, however, hepatic glycogen stores are depleted, and glucose is formed from noncarbohydrate precursors such as lactate, pyruvate, glycerol (derived from the backbone of triacylglycerols; see p. The formation of glucose does not occur by a simple reversal of glycolysis, because the overall equilibrium of glycolysis strongly favors pyruvate formation. Instead, glucose is synthesized by a special pathway, gluconeogenesis, which requires both mitochondrial and cytosolic enzymes. During an overnight fast, approximately 90% of gluconeogenesis occurs in the liver, with the remaining 10% occurring in the kidneys. However, during prolonged fasting, the kidneys become major glucose-producing organs, contributing an estimated 40% of the total glucose production. The most important gluconeogenic prescurors are glycerol, lactate, and the keto acids obtained from the metabolism of glucogenic amino acids. Glycerol Glycerol is released during the hydrolysis of triacylglycerols in adipose tissue (see p. Glycerol is phosphorylated by glycerol kinase to glycerol phosphate, which is oxidized by glycerol phosphate dehydrogenase to dihydroxyacetone phosphate, an intermediate of glycolysis. In the Cori cycle, bloodborne glucose is converted by exercising muscle to lactate, which diffuses into the blood.
Therefore depression or grief test cheap 300 mg eskalith, a series of biochemical concept maps have been created to depression glass buy cheap eskalith 300mg line graphically illustrate relationships between ideas presented in a chapter and to depression world history definition buy eskalith amex show how the information can be grouped or organized. Material is represented in a hierarchic fashion, with the most inclusive, most general concepts at the top of the map and the more specific, less general concepts arranged beneath. The concept maps ideally function as templates or guides for organizing information, so the student can readily find the best ways to integrate new information into knowledge they already possess. Concept boxes and links: Educators define concepts as "perceived regularities in events or objects. These broadly defined concepts are prioritized with the central idea positioned at the top of the page. The concepts that follow from this central idea are then drawn in boxes (Figure 1. The label on the line defines the relationship between two concepts, so that it reads as a valid statement, that is, the connection creates meaning. The lines with arrowheads indicate in which direction the connection should be read (Figure 1. Cross-links: Unlike linear flow charts or outlines, concept maps may contain crosslinks that allow the reader to visualize complex relationships between ideas represented in different parts of the map (Figure 1. Students learn to visually perceive nonlinear relationships between facts, in contrast to cross-referencing within linear text. Each amino acid also contains one of 20 distinctive side chains attached to the -carbon atom. The chemical nature of this R group determines the function of an amino acid in a protein and provides the basis for classification of the amino acids as nonpolar, uncharged polar, acidic (polar negative), or basic (polar positive). All free amino acids, plus charged amino acids in peptide chains, can serve as buffers. The -carbon of each amino acid (except glycine) is attached to four different chemical groups and is, therefore, a chiral, or optically active carbon atom. C represents the isoelectric point, or pI, and as such is midway between pK1 and pK2 for a nonpolar amino acid. The peptide would move to the cathode (negative electrode) during electrophoresis at pH 5. The two cysteine residues can, under oxidizing conditions, form a disulfide (covalent) bond. The net charge on the peptide at pH 5 is negative, and it would move to the anode. Given that the pKa of aspirin (salicylic acid) is 3, calculate the ratio of its ionized to un-ionized forms at pH 7. The linear sequence of the linked amino acids contains the information necessary to generate a protein molecule with a unique three-dimensional shape. The complexity of protein structure is best analyzed by considering the molecule in terms of four organizational levels: primary, secondary, tertiary, and quaternary (Figure 2. An examination of these hierarchies of increasing complexity has revealed that certain structural elements are repeated in a wide variety of proteins, suggesting that there are general "rules" regarding the ways in which proteins achieve their native, functional form. These repeated structural elements range from simple combinations of -helices and sheets forming small motifs, to the complex folding of polypeptide domains of multifunctional proteins (see p. Understanding the primary structure of proteins is important because many genetic diseases result in proteins with abnormal amino acid sequences, which cause improper folding and loss or impairment of normal function. If the primary structures of the normal and the mutated proteins are known, this information may be used to diagnose or study the disease. Peptide bond In proteins, amino acids are joined covalently by peptide bonds, which are amide linkages between the -carboxyl group of one amino acid and the -amino group of another. For example, valine and alanine can form the dipeptide valylalanine through the formation of a peptide bond (Figure 2. Peptide bonds are resistant to conditions that denature proteins, such as heating and high concentrations of urea (see p. Prolonged exposure to a strong acid or base at elevated temperatures is required to break these bonds nonenzymically.
Discount eskalith 300mg line. How to Control Anxiety Disorder.
See also specific type under Ig class switching and mood disorder dsm 4 code purchase 300 mg eskalith, 580 classes of depression symptoms diagnosis cheap eskalith 300mg otc, 577 depression era photos discount eskalith american express, 578t diseases caused by over- and underproduction of, 580 functions of, 578t, 579 genes for. See Michaelis constant Knockout genes, 400 Korsakoff psychosis, 473 674 Index Kozak consensus sequences, 361 Krabbe disease, 210t Krebs cycle. See also Metabolism Metabolic syndrome, 639 Metabolic pathway/metabolite flow, 133. See also specific micronutrients determination of requirements for, 465 minerals, 480, 480t digestion and absorption of, 462 vitamins. See Multispecific organic anion transporter Modeling, molecular, in protein structure analysis, 38 Molecular biology, 1. See Chaperones Molecular diagnostic tests, 4 Molecular docking programs, 38, 89 Molecular dynamics, 38 Molecular genetics, 1, 388. See also Cytochrome P450 system in metabolism of xenobiotics, 610 Monosaccharides, 113. See also specific type and Glucose absorption of, 460, 460f physiologic importance of, 115, 116t Monounsaturated fatty acids, 122, 123t.
However anxiety job 300 mg eskalith overnight delivery, vitamin D anxiety group order eskalith 300mg on-line, which is formed in the skin from 7-dehydrocholesterol on exposure to anxiety breathing problems order genuine eskalith on line sunlight, and niacin, which can be formed from the essential amino acid tryptophan, do not strictly comply with this definition. The -, -, and -carotenes and cryptoxanthin are quantitatively the most important provitamin A carotenoids. Although it would appear that one molecule of -carotene should yield two of retinol, this is not so in practice; 6 g of -carotene is equivalent to 1 g of preformed retinol. The total amount of vitamin A in foods is therefore expressed as micrograms of retinol equivalents. The intestinal activity of carotene dioxygenase is low, so that a relatively large proportion of ingested -carotene may appear in the circulation unchanged. Asterisk shows the site of cleavage of -carotene by carotene dioxygenase, to yield retinaldehyde. Any one cone cell contains only one type of opsin, and is sensitive to only one color. In the pigment epithelium of the retina, all-trans-retinol is isomerized to 11-cis-retinol and oxidized to 11-cis-retinaldehyde. This results in the release of retinaldehyde from the protein, and the initiation of a nerve impulse. The formation of the initial excited form of rhodopsin, bathorhodopsin, occurs within picoseconds of illumination. In deficiency, both the time taken to adapt to darkness and the ability to see in poor light are impaired. Retinoic Acid Has a Role in the Regulation of Gene Expression and Tissue Differentiation A major role of vitamin A is in the control of cell differentiation and turnover. Retinoid X receptors also form dimers with vitamin D, thyroid, and other a nuclear acting hormone receptors. The earliest sign of deficiency is a loss of sensitivity to green light, followed by impairment to adapt to dim light, followed by night blindness. More prolonged deficiency leads to xerophthalmia: keratinization of the cornea and blindness. Vitamin A also has an important role in differentiation of immune system cells, and even mild deficiency leads to increased susceptibility to infectious diseases. Also, the synthesis of retinol binding protein is reduced in response to infection (it is a negative acute phase protein), decreasing the circulating concentration of the vitamin, and further impairing immune responses. This undergoes a further reaction over a period of hours to form cholecalciferol, which is absorbed into the bloodstream. In temperate climates, the plasma concentration of vitamin D is highest at the end of summer and lowest at the end of winter. Ergocalciferol from fortified foods undergoes similar hydroxylation to yield ercalcitriol. In the liver, cholecalciferol is hydroxylated to form the 25-hydroxy-derivative, calcidiol. This is released into the circulation bound to a vitamin D binding globulin, which is the main storage form of the vitamin. In the kidney, calcidiol undergoes either 1-hydroxylation to yield the active metabolite 1,25-dihydroxy-vitamin D (calcitriol), or 24-hydroxylation to yield a probably inactive metabolite, 24,25-dihydroxyvitamin D (24-hydroxycalcidiol). Vitamin A Is Toxic in Excess There is only a limited capacity to metabolize vitamin A, and excessive intakes lead to accumulation beyond the capacity of binding proteins, so that unbound vitamin A causes tissue damage. Symptoms of toxicity affect the central nervous system (headache, nausea, ataxia, and anorexia, all associated with increased cerebrospinal fluid pressure); the liver (hepatomegaly with histologic changes and hyperlipidemia); calcium homeostasis (thickening of the long bones, hypercalcemia, and calcification of soft tissues); and the skin (excessive dryness, desquamation, and alopecia). Vitamin D Metabolism Is Both Regulated by and Regulates Calcium Homeostasis the main function of vitamin D is in the control of calcium homeostasis, and in turn, vitamin D metabolism is regulated by factors that respond to plasma concentrations of calcium and phosphate.

